The U.S. House Committee on Oversight and Reform held the second of a two-part hearing on Thursday titled “Examining JUUL’s Role in the Youth Nicotine Epidemic.” The Economic and Consumer Policy Subcommittee led the hearing as part of its investigation into “JUUL’s role in the youth nicotine addiction epidemic, marketing to youth, misleading health claims, and new partnerships with traditional tobacco companies.”
Panelists testifying in the second day of hearings included James Monsees, co-founder and Chief Product Officer of JUUL Labs, Inc.; Ashley Gould, Chief Administration Officer at JUUL Labs, Inc.; and Matthew Myers, the president of Campaign for Tobacco-Free Kids.
“While we made tremendous progress in preventing youth nicotine addiction at the start of the millenium, we face a much different challenge today,” Subcommittee Chairman Rep. Raja Krishnamoorthi, D-IL, said in his opening statements.
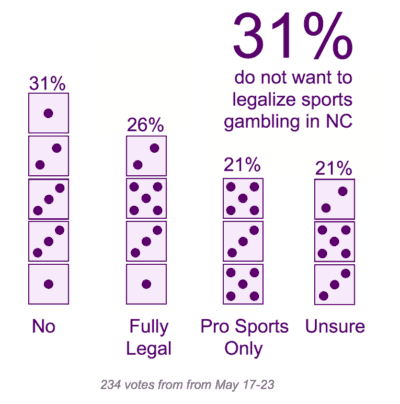
According to the 2018 National Youth Tobacco Survey, 3.6 million high school and middle school students used e-cigarettes, an increase of 1.5 million in one year.
“The ability for kids to get addicted to e-cigarettes, and the inability for adults to prevent it, poses an unparalleled and unprecedented challenge,” Krishnamoorthi said.
In follow-up statements, Rep. James Comer, R-KY, agreed that no one wants kids using tobacco products or vaping, however, he brought into question the committee’s hearing of Juul Labs, Inc.
“What concerns me about this hearing is, once again, this committee is using its oversight authority to bring in another private company and question them about their business,” Comer said.
Comer also expressed concerns that regulations of e-cigarettes like Juul could keep smokers from switching to a “positive public health alternative.”
“Prematurely over-regulating companies such as Juul could have a disastrous effect that impedes access to one of the most successful products for smoking cessation,” Comer said. The topic of Juul and smoking cessation would be a key point of discussion throughout the hearing.
Monsees, in his witness statement, said that he and co-founder Adam Bowen started Juul Labs as smokers who were looking for a way to quit.
“For us, giving up smoking required an alternative which did not exist at the time. We founded Juul Labs to invent one,” Monsees said. “The product we developed holds the promise to do what no previous technology has done — help adult smokers stop smoking combustible cigarettes in a widespread and consistent basis.”
“Mr. Chairman, put simply, Juul Labs isn’t Big Tobacco,” Monsees later said. “We are here to eliminate its product: the cigarette.”
In questioning, some members of Congress took aim at these statements, and in particular, at Juul’s promotion of itself as a quitting device while not being FDA-approved as a smoking cessation product.
Debbie Wasserman Schultz, D-FL, introduced the Preventing Opportunities For Teen E-Cigarette and Tobacco Addiction (PROTECT) Act earlier this year, and recognized Juul Lab’s support of the proposed legislation.
“I want to take your statements today in good faith, that you’re taking the epidemic of youth e-cigarette use seriously,” Wasserman Schultz said. “But unfortunately, there is so much evidence to the contrary.”
She noted the nicotine content levels of Juul products, which were three times higher (at 59mg/mL) than other e-cigarette products, “making Juul the most potent e-cigarette on the market.” She also pointed that the E.U. and U.K. put a cap on nicotine product levels in their market at 20 mg/mL, and Juul Labs sells them a product meeting their required levels.
“So obviously Juul has determined that it can still make money selling a product at less dangerous nicotine levels. So, yes or no, do you think that at 20 milligrams per milliliter Juul can accomplish your stated mission of helping adult smokers to quit smoking conventional cigarettes?” Wasserman Schultz asked Monsees.
“We would love to try,” Monsees said.
“Okay, but you’re already trying in every country except this one,” Wasserman Schultz said.
During questioning, Wasserman Schultz also said, “You appear to be marketing yourself as a smoking cessation device, however you are not FDA-approved for that purpose and efforts to increase nicotine delivery highlight why that is the case. FDA-approved smoking cessation products include instructions for stepping down nicotine intake and for weaning off nicotine entirely. Mr. Monsees, does your product come with these instructions?”
“It does not, and we would never market it as a cessation product,” Monsees responded. “Let me be very clear, Juul is not a cessation product.”
“That’s news to me and probably everyone in this room that Juul is not marketing yourselves as a nicotine or a smoking cessation product. So that’s every other vaping product except yours?” Wasserman Schultz said, visibly frustrated.
Rep. Rashida Tlaib, D-MI, continued asking Monsees about Juul’s product promotion for smokers to quit cigarettes while showing an ad which told smokers to “make the switch” to Juul.
“You’re trying to get around FDA regulations, Mr. Monsees,” Tlaib said. “The FDA regulates smoking cessation devices, the things that actually help people quit smoking, but Juul has avoided explicitly calling itself a cessation device, which Juul believes helps it avoid this particular FDA regulation. … Mr. Monsees, I imagine you chose your words very carefully in these ads. Yes or no, did your lawyers tell you that this ad was not claiming to be a therapeutic cessation device?”
“We do not claim for Juul to be a cessation product, and let me tell you exactly why,” Monsees said. “The history of cessation products have extremely low efficacy. That’s the problem we’re trying to solve here. So if we can give consumers an alternative and market it right next to other cigarettes, then we can actually make something work.”
“You targeted children to do that, too,” Tlaib interjected.
Tlaib later asked, “If you made the direct statement to the U.S. government that your products were smoking cessation devices, or what you’ve been telling these other folks, healthier than cigarettes, you would be expected to be regulated that day wouldn’t you?”
“I can’t state more emphatically, Juul is specifically and on purpose not a cessation product,” Monsees responded.
In the second panel, Ashley Gould, Chief Administration Officer at Juul Labs, also experienced questioning on the topic of Juul as a cessation device.
“Ms. Gould, why has Juul never conducted a clinical trial to prove the number one claim that it makes — that its devices help adults quit cigarettes?” Chairman Krishnamoorthi asked.
Gould responded that the company had undertaken a number of studies, though it has not conducted clinical trials.
EducationNC’s recap of the first part of the hearings can be found here. For more on youth e-cigarette use, follow our series here.
Recommended reading